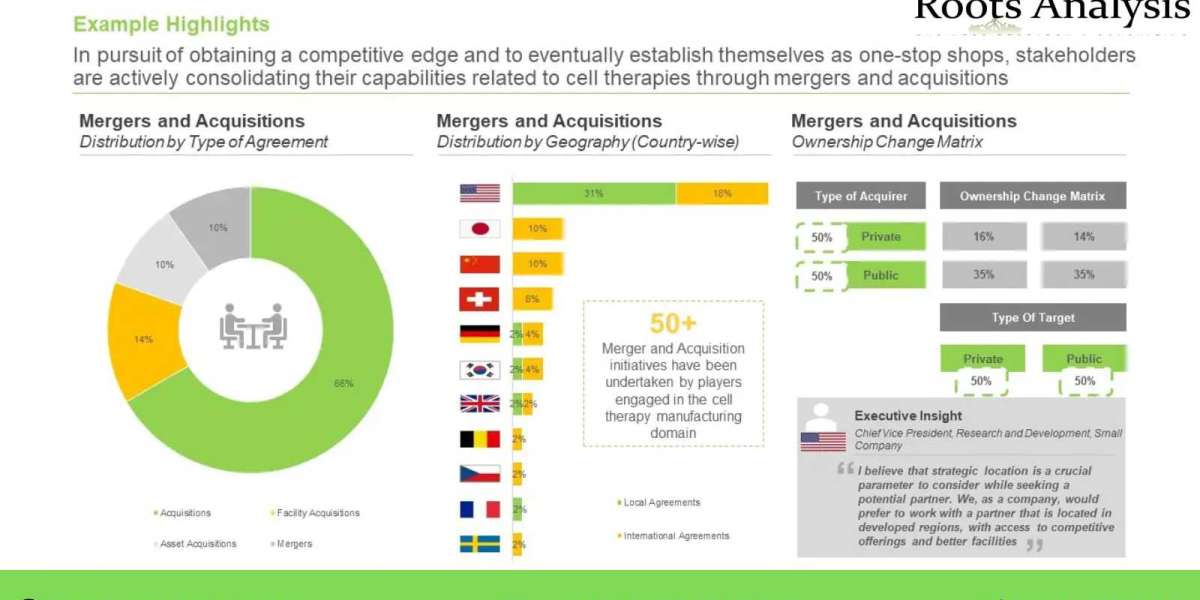
The focus of stakeholders has now shifted to optimizing the cell therapies manufacturing process. Over the years, several advanced and innovative automation tools and technologies have been developed; these have been demonstrated to hold the potential for significant reduction in the cost associated with the manufacturing of advanced therapy medicinal products, thereby, making such products more affordable. One such emerging concept, namely GMP-In-A Box, offers several advantages, including increased throughput, decreased idle time between batch runs and reduced manual labor.
For the success of cell-based therapies, an effective manufacturing platform and a robust supply chain model is imperative. It is worth mentioning that a sustainable supply chain can actually enable further reductions in the cost of goods and, in turn, prices of such therapies.
Unlike conventional pharmaceuticals, cell therapies require a circular supply chain model. For instance, in case of an autologous cell therapy; the first step involves the procurement of input material (cells) from the patient, since these therapies are developed using the patient’s own cells. The material is then sent to a manufacturing facility and finally, needs to be shipped back to the location of the patient for administration.
However, there are several hurdles associated with the transition of cell-based therapy products from laboratory to clinical scale, such as regulatory compliance, lack of advanced facilities and infrastructure. Moreover, personnel with high levels of technical expertise and scientific acumen became a prerequisite. In addition, the technology platforms used for the development of such advanced therapy medicinal products (ATMPs) need to be approved by regulatory authorities.
Owing to these inherent complexities associated with manufacturing such products, scaling up their production to the commercial level is considered as one of the major challenges in this domain.
Despite the challenges associated with the development and production of such therapies, we believe, that the benefits offered by them outweigh the hurdles. They are likely to serve as important drivers of the industry’s growth. Efforts to introduce automation technologies in cell therapy manufacturing are underway, and if implemented successfully, can significantly help in the elimination of human intervention and reduce the risk of contamination.
As a consequence, it is likely to result in a marked increase in product consistency, ensure the maintenance of sterility, and decrease the production time and cost. In a nutshell, cell therapies are expected to soon represent one of the prominent therapeutic options within the mainstream healthcare.
For additional details, please visit https://www.rootsanalysis.com/blog/cell-therapies-manufacturing-automation/ or email [email protected]
You may also be interested in the following titles:
- Smart Labels Market: Industry Trends and Global Forecasts, 2022-2035
- AI-based Digital Pathology / AI Pathology Market: Industry Trends and Global Forecasts, 2022-2035
About Roots Analysis
Roots Analysis is a global leader in the pharma / biotech market research. Having worked with over 750 clients worldwide, including Fortune 500 companies, start-ups, academia, venture capitalists and strategic investors for more than a decade, we offer a highly analytical / data-driven perspective to a network of over 450,000 senior industry stakeholders looking for credible market insights.
Contact:
Ben Johnson
+1 (415) 800 3415
[email protected]